Japanese Journal of Gastroenterology Research
Research Article - Open Access, Volume 1
The role of the area postrema in belching
*Corresponding Author: Ivan M Lang, DVM, PhD
MCW Dysphagia Research Laboratory, Division of Gastroenterology and Hepatology, Department of Medicine, Medical College of Wisconsin, 8701 Watertown Plank Road, Milwaukee WI 53226, USA
Tel: 414 456-8138
Email: imlang@mcw.edu
Received : Apr 28, 2021
Accepted : May 21, 2021
Published : May 25, 2021
Archived : www.jjgastro.com
Copyright : © Lang IM (2021).
Abstract
Introduction: The primary reflex involved with belching is associated with the activation of neurons in the area postrema (AP), therefore, we investigated the role of the AP in the activation of belching.
Methods: The effects of mechanical lesions of the dorsal brainstem on activation of belching, esophago-UES contractile reflex (EUCR), and the pharyngeal swallow (PS) were determined in 13 decerebrate cats.
Discussion: Bilateral lesions of the dorsal brain stem which included the rostral AP significantly (P< 0.05, N = 7) blocked belching, but not the other reflexes. Bilateral lesions of the rostral AP only blocked belching (N=2). When belching was blocked, the same belch stimulus activated EUCR (in 6 out of 7 animals). Unilateral lesions of the AP (N = 3) did not block belching.
Conclusions: The rostral AP is essential for activation of belching. We hypothesize that the AP serves an inhibitory function to prevent reflexes that promote orthograde transport, e.g. PS, and reflexes that prevent supra-esophageal reflux, e.g. EUCR, thereby, facilitating retrograde transport.
Keywords: Belching; brain stem; area postrema.
Citation: Lang IM. The role of the area postrema in belching. Japanese J Gastroenterol Res. 2021; 1(1): 1005.
Introduction
Belching has been studied for many years [1-3], however, while the sensory and motor mechanisms have been investigated [1], the central nervous system control of belching has not been determined. Belching can be activated in the decerebrate animal [4], therefore, the control centers for belching must be in the brain stem.
Athough the brain stem sites controlling belching have not been identified, one study [5] identified the brain stem nuclei activated during the primary reflex that comprises belching, i.e. the esophago-UES relaxation reflex (EURR) [6]. This study [5] found that stimulation of EURR was associated with a significant increase in c-fos activation in both sensory and motor nuclei of this reflex, but also the area postrema (AP).
Belching, like vomiting [7,8], is a complex response composed of separate reflexes of multiple organs that occur in a very organized fashion [1]. Although there is controversy whether a vomiting center exists or what brain nuclei comprise the vomiting center [9-11], ablation of the AP has been shown to block various aspects of the vomiting process [9-11]. Considering that the AP has a significant role in emesis [9-11] and that the AP is significantly activated during belching [5], it is hypothesized that the AP has a significant role in the belch process as it does in the vomiting process. That is, it is hypothesized that the AP is not just activated during belching, but that its integrity is essential for the operation and function of belching.
Materials and Methods
These studies were conducted using seven male and six female domestic short hair cats weighing 2.8 to 4.5 kg which were ten to fourteen months old and obtained from Liberty Research Inc. All studies were conducted with the approval of the Animal Care and Use Committee of the Medical College of Wisconsin.
Animal preparation
The cats were decerebrated to allow investigation in a minimally anesthetized and tranquilized animal model to study a function controlled by the brain stem. The cats were fasted overnight and decerebrated the next morning. The animals were anesthetized (3% isofl urane), the ventral neck region exposed, the trachea intubated, and the carotid arteries ligated. The skull was exposed, and a hole over a parietal lobe was made using a trephine. The hole was enlarged using rongeurs, the central sinus was ligated and cut, and the brain was severed midcollicularly. The forebrain was then suctioned out of the skull, and the blood vessels of the Circle of Willis were coagulated by suction through cotton balls soaked in warm saline. The boney sinuses were fi lled with bone wax, the exposed brain was covered with paraffi n oil-soaked cotton balls, and the skin over the skull was sewn closed. The animals were then placed supine on a heating pad (Harvard Homeothermic monitor), and the body temperature maintained between 38 and 40oC.
After decerebration the larynx and pharynx were exposed and the trachea cannulated. EMG electrodes were placed on the geniohyoideus (GH), thyrohyoideus (TH), cricothyroideus (CT), and cricopharyngeus (CP). The CP is the primary muscle of the UES, and its actions are considered responses of the UES [12]. The abdomen was then opened along the ventral midline and a fi stula of the proximal stomach was formed using a 3 ml plastic syringe that exited the abdominal cavity. This fistula was used to drain the stomach of gastric acid and to insert stimulation devices into the esophagus without disturbing the pharynx or larynx and a manometric catheter to record esophageal motility. The femoral vein was cannulated for infusion of saline, and the femoral artery cannulated to record arterial blood pressure.
The head of the animal was then placed in a stereotaxic apparatus (Kopf Instruments) and a heating pad was placed under the body and the body temperature maintained between 38-40oC. The brain stem was then exposed by the removal of the cisterna magna using rongeurs and cutting the dura mater. Warm mineral oil was placed onto the brain stem to keep it moist and the head was oriented such that the brain stem was parallel with the ground to prevent the oil from running out of the exposed brain stem. At the end of the study the animal was euthanized (Beuthanasia, 1 ml/kg IV) and the brain stem removed and placed in 10% formalin.
Experimental protocol
Studies were begun two to three hours after the surgical preparation. The control study for each cat consisted of determining the threshold stimulus for three reflexes: 1) belching, 2) esophago-UES contractile reflex (EUCR), and 3) pharyngeal swallow (PS). Each reflex was activated at least three times 5 to 10 minutes apart to obtain the threshold stimulus magnitude for each reflex. After obtaining the control studies, the brain stem was lesioned, as described below, which took 15 to 30 minutes. The cat was then allowed 30 to 60 minutes to recover from the lesion and the reflexes were tested again in the same order as during the control studies. We used up to three times the original threshold stimulus magnitude to test the ability of the reflex to be activated.
The effects of the lesions on blood pressure were also tested. The control blood pressure was obtained just before the lesion and the experimental blood pressure was obtained after the lesion just before retesting of the reflexes.
Data acquisition and storage
Recording techniques
Electromyography (EMG): Bipolar Teflon-coated stainless steel wires (AS 632: Cooner Wire, Chatsworth, CA) bared for 2-3 mm were placed in each muscle and the wires were fed into differential amplifiers (A-M Systems 1800). To record muscle activity bilaterally, each pole of the EMG recording was placed in each side of the muscle. The electrical activity was filtered (bandpass of 0.1-3.0 KHz) and amplified (1,000-10,000 times) before feeding into the computer.
Manometry: Blood Pressure and Esophageal Motility: Blood pressure was recorded using a Statham pressure transducer. Esophageal motility was recorded using a three channel (3 cm apart) solid state manometric catheter (Gaeltec, Medical Measurements Inc). The manometry output signals were fed into low level DC preamplifiers (Grass P122) set at 3 Hz high frequency cutoff filtration. The manometric signals were stored on computer.
Computer data acquisition: All data was acquired (1800 Hz) and analyzed using Dataq Instruments data acquisition hardware and software.
Stimulation techniques
Three reflex responses were tested: belch, esophago-UES contractile reflex (EUCR), and the pharyngeal swallow (PS).
Belch: The belch was stimulated by rapid injection of air into the esophagus. The belch consists of activation of the esophago-UES relaxation reflex (EURR) concomitant with activation of thyrohyroideus and cricothyroideus [13]. A 2 mm diameter catheter with a side hole at its tip was inserted through the gastric fistula. The side hole allowed injected air to be directedat right angles to the esophageal mucosa. The catheter was attached to a pneumatic pump (PicoPump, World Precision Instruments) which injected air at 4 -15 PSI for 0.5s.
EUCR: The esophago-UES contractile reflex (EUCR) was activated by the slow injection of air into the esophagus through the same tube used for stimulation of belching. Air (2 to 5 ml) was injected by hand from a 5 ml syringe.
Pharyngeal Swallow: Water (0.5 to 1.5 ml) was injected by hand into the pharynx through the oral cavity.
Brain stem techniques
Brain stem lesions: A blunted 15 gauge metal needle was attached to vacuum source and positioned by hand above the dorsal surface of the brainstem targeting the AP. The advantage of this method is that the boundaries of the lesions were easily determined histologically, similar to a knife cut. In addition, finding the target nucleus, i.e., the AP, did not require stereotaxic coordinates as the AP was readily visible on the dorsal surface of the brain stem. Various amounts of AP and other dorsal brain stem nuclei were suctioned from the brainstem. It was difficult to remove only the AP, but it was not necessary as it was also desired to lesion other nearby structures for statistical comparison to distinguish the effects of the AP removal from the removal of other dorsal brain stem nuclei.
Histology: After a few days of fixation in formalin, the brain stems were sliced in a microtome at 40 μm, the sections placed on a slide, and the slices stained using a Nissl (crestyl violet) staining procedure.
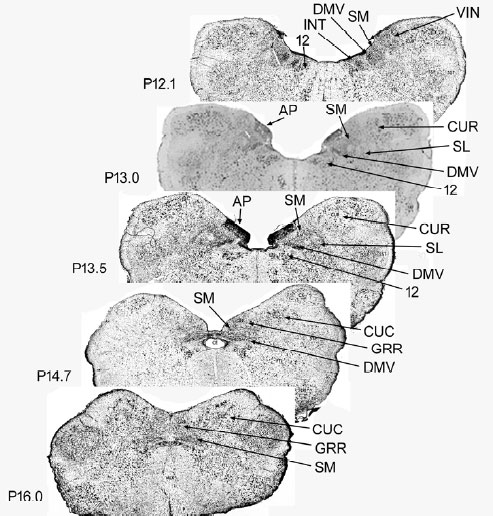
Brain stem lesion identification: The identification of the brain stem nuclei removed were determined by comparing the histological sections of the experimental animals with sections from cat stereotaxic atlases (Figure 1) [14,15].
Statistical Methods
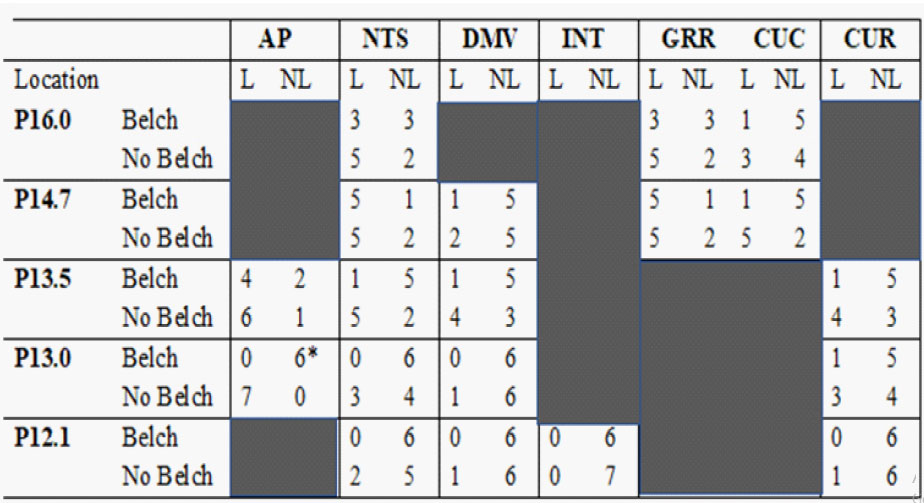
The lesions either blocked or did not block belching and a known nucleus was either lesioned or not, therefore, we tested the effectiveness of lesions of particular nuclei in blocking belching using the Fisher's Exact test. All of the lesions were of the dorsal surface of the brain stem in the region of the AP between coordinates P16.0 to P12.1 of the Clarke [16] frontal zero plane. We tested the effectiveness of lesions of the following brain stem nuclei which were within this region: AP, nucleus tractus solitarius (NTS) which includes the medial (SM) and lateral (SL) subnuclei, dorsal motor nucleus of the vagus (DMV), rostral division of the gracile nucleus (GRR), nucleus intercalatus (INT), caudal division of the cuneate nucleus (CUC), rostral division of the cuneate nucleus (CUR), and hypoglossal nucleus [12]. The effects of the brain stem lesions on systolic, diastolic, and mean blood pressures were determined using the paired Student's t-test. A P value of < 0.05 was considered statistically significant.
Results
Brain stem techniques
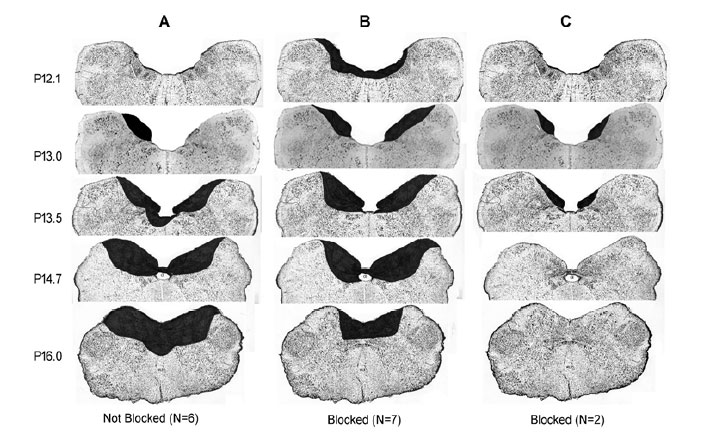
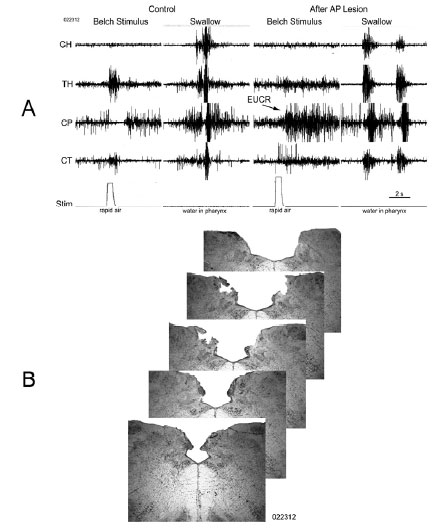
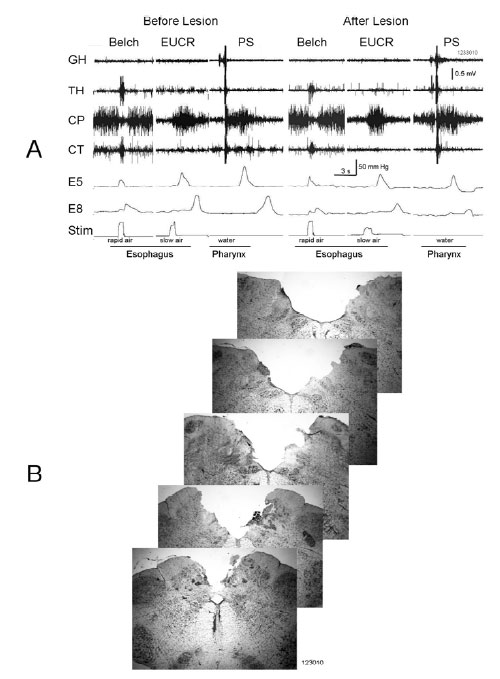
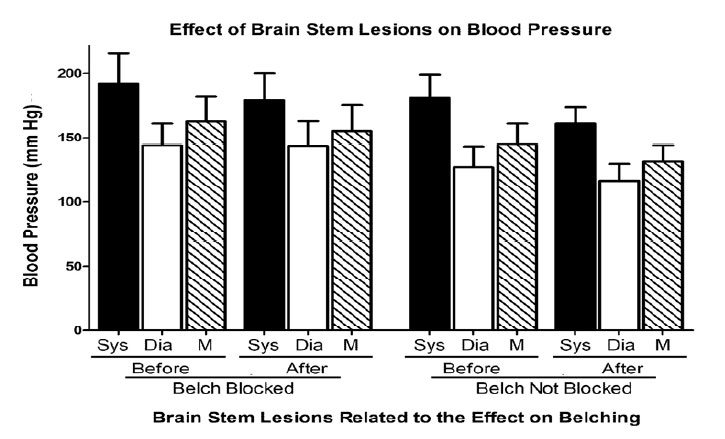
Bilateral lesions of the dorsal brain stem,which included the AP at P13.0 significantly (P < 0.05) blocked the belch response compared to dorsal brain stem lesions which included brain stem nuclei, e.g., NTS and DMV caudal to P12.1, as well as unilateral lesions of the AP (Table 1, Figure 2). Bilateral lesions, in which the rostral AP was the only nucleus lesioned (N = 2), blocked belching (Figures 2 and 3). Three of the lesions, which did not block belching, included unilateral lesions of the rostral AP and an example of the effects of such a lesion are depicted in Figure 4. Lesions, which included the NTS at P13.5, came close to significance (P = 0.10) for blocking belching (Table 1).
Lesions of the AP, which blocked belching, had no significant effect (7 out of 7 animals, P = 1.0) on activation of EUCR or PS (Figure 3). In addition, when lesions of the AP blocked belching (N=7) that same stimulus, used to activate belching, now activated EUCR (N = 6, Figure 3).
Effects of brain stem lesions on blood pressure
Whether the brain stem lesions blocked the AP or not, these lesions had no significant effect (P > 0.05 for all comparisons) on blood pressure (Figure 5), even though in many cases other dorsal brain stem nuclei, e.g., NTS, DMV, etc., were also lesioned (Table 1, Figures 2 and 3).
Table 1: Effect of Brain Stem Lesions on Blocking Belch Response
Discussion
These studies show that bilateral lesions of the brain stem which included the rostral portions of the AP significantly blocked the activation of the belch response compared with lesions which included the same dorsal brain stem nuclei, e.g. NTS, DMV, GRR, INT, CUC, CUR, and 12, but did not include the rostral AP bilaterally. These results strongly suggest that the integrity of the rostral AP bilaterally is necessary for activation of belching. This was confirmed as bilateral lesions of only the rostral AP blocked belching. Therefore, while prior studies (5) found that belching is associated with increased activation of neurons in the AP, the AP is not just activated during belching its integrity is essential for the activation of belching.
Lesions of the dorsal brain stem had no effect on blood pressure whether belching was blocked or not, therefore, it is unlikely that the effects on belching are secondary to changes in cardiovascular function. In addition, these lesions had no significant effect on activation of EUCR or PS. Therefore, it is unlikely that the effects of AP lesions on belching are secondary to alterations in other functions.
Considering the current findings and that 1) the belch response is a complex reflex involving multiple sub-reflexes that requires a control center [1], 2) the sensory and motor nuclei of the sub-reflexes of the belch response are not in the AP [17,18], and 3) a prior study found that the AP is significantly activated during belching [5], one might conclude that the rostral AP may be the brain stem motor control center for the belch response. However, a study [19] using similar techniques concluded the same thing regarding emesis seventy years ago which has subsequently been challenged by many studies.Lesions of the AP block some, but not all types of emesis [9-11]. Emesis induces activation of c-fos in the AP, but also in nuclei of the ventrolateral reticular formation of the brain stem [20]. More significantly, it was found that a non-respiratory set of neurons of the NTS receive convergent input from the AP and vagus nerve which project to the Botzinger nucleus [21], which is in the ventrolateral reticular formation. Stimulation of the Botzinger nucleus has been found to elicit retching and vomiting, even after lesions of the NTS [22]. Thus, the most recent studies suggest that the brain stem motor control center for retching and vomiting is not the AP, and therefore, the AP may also not be a motor control center for belching.
If the AP is not the belching motor control center, then what is its function? The AP has been shown to have many functions especially related to cardiovascular control [23], but given the lack of effect of the brain stem lesions on blood pressure in our study, it is highly unlikely that disruption of these cardiovascular functions had any role in the blockade of belching. However, there is one function of the AP that may be related to belching and that is vomiting [11].
Both vomiting [8] and belching [1] function to move gastric contents, fluid or air, out of the body in a retrograde fashion. In addition, the reflexes involved in belching have been shown to be involved in gastro-esophageal and esophago-pharyngeal reflux, as discussed below. Therefore, the function of both vomiting and belching is opposite the function of other upper digestive tract reflexes, e.g. swallowing [1,8], EUCR [6], etc. The function of these other reflexes is either to promote orthograde transport, e.g., swallowing, or to prevent supra-esophageal reflux, e.g., EUCR. Some of the stimuli that activate belching or vomiting are also capable of activating some of these other reflexes, as shown in this and other studies [1,6]. If both types of reflexes were activated simultaneously significant functional problems would result. Therefore, it is essential during belching and vomiting that the reflexes that promote orthograde transport or prevent supra-esophageal reflux be inhibited.
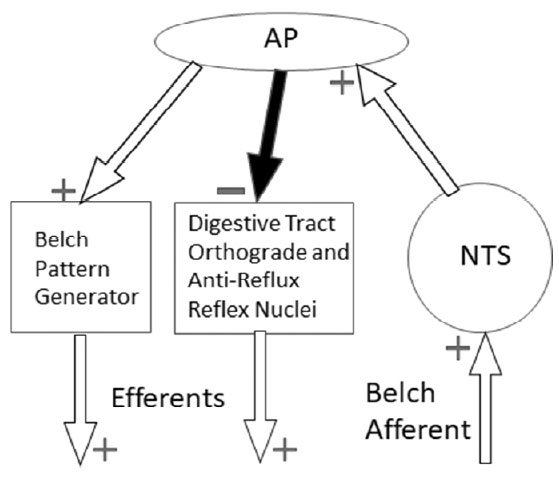
Perhaps the AP serves as a central motor inhibitory center. We observed such an effect in our current studies. We found that when the AP was lesioned, such that belching was blocked, the very same belch stimulus activated the EUCR. That is, during the activation of belching the EUCR, i.e., a reflex that prevents supra-esophageal reflux [6], must have been inhibited. The inhibitory nature of the AP on digestive tract functions is supported by experimental studies. Lesions of the AP in rats eliminated the initial inhibition of distal gastric motility stimulated by the emetic agent, apomorphine [24], and electrical or chemical stimulation of the AP inhibited intragastric pressure in rats [25]. It is therefore hypothesized that a function of the AP in belching, and perhaps vomiting, is not as a motor control center for a retrograde response, but as an inhibitory center that permits the retrograde event to occur by inhibiting reflexes that promote orthograde transport or prevent supra-esophageal reflux.
Studies suggest that lesions of the AP block vomiting because the lesion transectsa neural connection between the AP and NTS [11]. It is possible that the lesions of the AP also transected a neural pathway with the NTS to block belching, as the NTS is adjacent to the AP [17,18] and it was found that brain stem lesions of the NTS at P13.5 were almost significantly (P= 0.10, N=6) related to blocking of belching. It is possible this effect would have been statistically significant if a higher N had been used. However, if AP lesion blocked belching by transecting an NTS pathway this would support the inhibitory hypothesis for the role of the AP in belching. If the role of the AP is as an inhibitor of reflexes that promote orthograde transport or prevent supra-esophageal reflux, then it must receive a sensory input from the NTS. Therefore, a neural projection from the NTS to the AP would be necessary and would explain the ability of AP lesions to block belching and vomiting without the AP being a motor control center for either response (Figure 6). There must be an important physiological reason for this neural projection from NTS to AP, which does not occur with other reflexes, and it might be the necessity of an inhibitory response during belching and vomiting.
Another feature of the effects of AP lesions on vomiting fits the hypothesis that the AP is an inhibitory rather than a motor control center. It has been observed that not all forms of vomiting are blocked by AP lesions [9-11]. Vomiting due to radiation-induced stimulation of vagal afferents [26-28] and chemical stimulation of the CTZ [29], but not motion sickness [4], are blocked by AP lesions. That is, vomiting which needs to remove contents from the digestive tract, i.e. CTZ and vagal afferent activation, require the AP, whereas types of vomiting that do not require expulsion from the digestive tract, i.e., motion sickness, do not. Chemical activation of the CTZ occurs after ingestion of poisonous substances that get absorbed [29] and vagal activation occurs after ingested substances irritate the digestive tract mucosa [2]. If as hypothesized that activation of the AP is needed for digestive tract expulsion, i.e. for vomiting and belching, and in order to accomplish this the digestive tract reflexes that promote orthograde transit or prevent supra-esophageal reflux must be inhibited, this would explain why only processes, i.e. vomiting and belching, designed to empty the digestive tract require the AP.
While belching may not be a significant clinical event, the reflexes that comprise belching are the same reflexes that are involved indigestive tract reflux. The transient relaxation of the lower esophageal sphincter (TLESR) is involved in gastroesophageal reflux (GER) [30,31], and it is the initial reflex that initiates the belch response [2,32]. Transient relaxation of the upper esophageal sphincter (TUESR) occurs during esophago-pharyngeal reflux (EPR) [33,34], and TUESR is the primary reflex of belching [2,6]. Therefore, given that the AP has a significant role in belching, it is likely that the AP may also have a significant role in the generation or control of GER and EPR.
A limitation of this study was the lesion technique. There are basically two lesion techniques, i.e., mechanical and chemical [7,9-11,25,28]. Chemical techniques have the advantage that some can be reversed and some specifically block only synapsesor certain types of synapses. Blocking only synapses can distinguish neuronal activation from axonal transmission. The mechanical techniques, knife cuts or suction, have the advantage of more accurate delineation of the limits of the lesion. Considering that the AP is not a nucleus that contains many axonal pathways [11, 25, 28], and it was needed to first know whether the AP was involved in belch before attempting to define the neurochemical nature of this effect, it was decided that a mechanical lesion technique was most appropriate. Also, suction rather than knife cut was used because the AP is on the dorsal surface of the brain stem and the brain stem is surrounded by bone on three sides. This anatomy provides little access to lesion the dorsal surface using a knife. Therefore, suction was used to mechanically lesion just the dorsal surface of the brain stem.
A consequence of the above limitation as well as the nature of the experimental animal, i.e. cat, was the low N value of one of the findings. While statistical probability using the Fisher Exact test revealed (P < 0.05, N = 13) that the primary nucleus involved in activating the belch response was the rostral AP, I was able to bilaterally lesion only the rostral AP in two animals. Given the rate of success of this procedure, it might have required experimentation on twenty-six addition animals to obtain statistical significance for this procedure. The unnecessary killing of large animals, e.g., cats and dogs, is against Public Health Service (PHS) policy. The PHS policy of Humane Care and Use of Animals requires that for research purposes only "the minimum number required to obtain valid results" be used. It was concluded that this criterion had ben met.
Conclusion
In summary, bilateral lesions of the rostral portion of the area postrema block belching, and therefore, the AP contains the brain stem neural circuitry needed for the activation of the reflexes that comprise the belch. The belch response inhibits the activation of a reflex that prevents reflux, i.e. EUCR; and lesions of the AP do not block pharyngeal swallowing or activation of the EUCR. Therefore, it is hypothesized that the AP functions in belching as an inhibitory center that prevents the activation of reflexes that promote orthograde transit or prevent supra-esophageal reflux.
Acknowledgements: The support and financial assistance of Dr. Reza Shaker and technical assistance of Dr. Bidyut K. Medda were greatly appreciated.
References
- Lee YC, Lee YL, Chuang JP, Lee JC. Differences in survival between colon and rectal cancer from SEER data. PloS One. 2013; 8: e78709
- Folkman J. Role of angiogenesis in tumor growth and metastasis. Semin Oncol. 2002; 29: 15-18.
- Rmali KA, Puntis MC, Jiang WG. Tumour-associated angiogenesis in human colorectal cancer. Colorectal Dis. 2007; 9: 3-14.
- Koch B, Giedl J, Hermanek P, Kalden JR. The analysis of mononuclear cell infiltrations in colorectal adenocarcinoma. J Cancer Res Clin Oncol. 1985; 109: 142-151.
- Zhang Y, Sun M, Huang G, Yin L, Lai Q, Yang Y, et al. Maintenance of antiangiogenic and antitumor effects by orally active low-dose capecitabine for long-term cancer therapy. Proc Natl Acad Sci U S A. 2017; 114: E5226-E5235.
- Yamazaki K, Nagase M, Tamaga H, Ueda S, Tamura T, Murata K, et al. Randomized phase III study of bevacizumab plus FOLFIRI and bevacizumab plus mFOLFOX6 as first-line treatment for patients with metastatic colorectal cancer (WJOG4407G). Ann Oncol. 2016; 27: 1539-1546.
- Cunningham D, Lang I, Marcuello E, Lorusso V, Ocvirk J, Shin DB, et al. Bevacizumab plus capecitabine versus capecitabine alone in elderly patients with previously untreated metastatic colorectal cancer (AVEX): an open-label, randomised phase 3 trial. Lancet Oncol. 2013; 14: 1077-1085.
- Tabernero J, Yoshino T, Cohn AL, Obermannova R, Bodoky G, Garcia-Carbonero R, et al. Ramucirumab versus placebo in combination with second-line FOLFIRI in patients with metastatic colorectal carcinoma that progressed during or after first-line therapy with bevacizumab, oxaliplatin, and a fluoropyrimidine (RAISE): a randomised, double-blind, multicentre, phase 3 study. Lancet Oncol. 2015; 16: 499-508.
- Van Cutsem E, Joulain F, Hoff PM, Mitchell E, Ruff P, Lakomý R, et al. Aflibercept Plus FOLFIRI vs. Placebo Plus FOLFIRI in Second-Line Metastatic Colorectal Cancer: a Post Hoc Analysis of Survival from the Phase III VELOUR Study Subsequent to Exclusion of Patients who had Recurrence During or Within 6 Months of Completing Adjuvant Oxaliplatin-Based Therapy. Target Oncol. 2016; 11: 383-400.
- Zhao HY, Ooyama A, Yamamoto M, Ikeda R, Haraguchi M, Tabata S, et al. Molecular basis for the induction of an angiogenesis inhibitor, thrombospondin-1, by 5-fluorouracil. Cancer Res. 2008; 68: 7035-7041.
- Shi H, Jiang J, Ji J, Shi M, Cai Q, Chen X, et al. Anti-angiogenesis participates in antitumor effects of metronomic capecitabine on colon cancer. Cancer Lett. 2014; 349: 128-135.
- Uboha N, Hochster HS. TAS-102: a novel antimetabolite for the 21st century. Future Oncol. 2016; 12: 153-163.
- Fox SB, Moghaddam A, Westwood M, Turley H, Bicknell R, Gatter KC, et al. Platelet-derived endothelial cell growth factor/thymidine phosphorylase expression in normal tissues: an immunohistochemical study. J Pathol. 1995; 176: 183-190.
- Gottfried E, Kunz-Schughart LA, Weber A, Rehli M, Peuker A, Müller A, et al. Expression of CD68 in non-myeloid cell types. Scand J Immunol. 2008; 67: 453-463.
- Danilov SM, Metzger R, Klieser E, Sotlar K, Trakht IN, Garcia JGN. Tissue ACE phenotyping in lung cancer. PLoS One. 2019; 14: e0226553.