Japanese Journal of Gastroenterology Research
Case Report - Open Access, Volume 1
A solitary granular cell tumor of stomach: A rare entity
Rachana Chaturvedi1*; Pooja Naik2
1Associate Professor (GI & Liver Division), Seth G.S. Medical College & KEM Hospital, Mumbai India.
2Seth G.S. Medical College & KEM Hospital, Mumbai, India.
*Corresponding Author: Rachana Chaturvedi
Associate Professor (GI & Liver division), Seth G.S. Medical College & KEM Hospital, Mumbai, India.
Email: rachanachaturvedi@yahoo.co.in
Received : Sep 23, 2021
Accepted : Nov 08, 2021
Published : Nov 10, 2021
Archived : www.jjgastro.com
Copyright : © Chaturvedi R (2021).
Introduction
Granular cell tumor (GCT) is uncommon, originates from Schwann cell and is usually benign. It is most commonly seen in skin and subcutaneous tissue of chest and upper extremity, tongue, breast, female genital tract and respiratory tract. Involvement of Gastrointestinal Tract (GIT) is seen only in 8% of cases and still rarer in stomach; more frequent in esophagus followed by colon. Most of GCTs are reported to be synchronously associated with the similar tumor at esophagus or other gastrointestinal location [1]. Here we report a rare case of solitary GCT of stomach.
Citation: Chaturvedi R, Naik P. A solitary granular cell tumor of stomach: A rare entity. Japanese J Gastroenterol Res. 2021; 1(7): 1033.
Case report
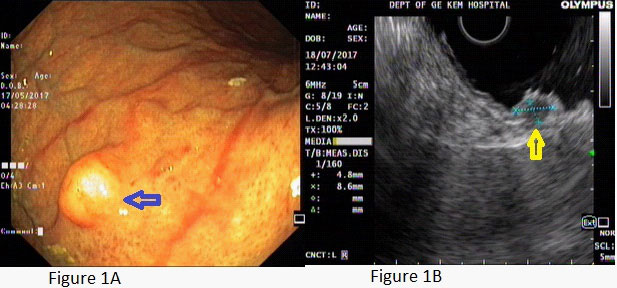
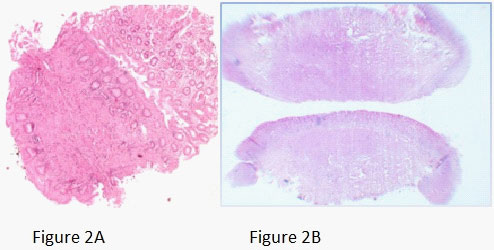
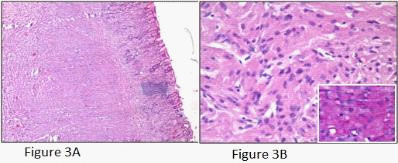
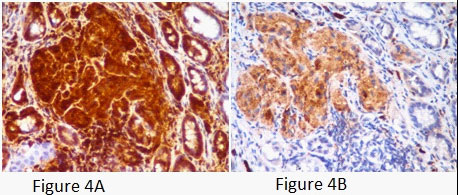
A young female presented with postprandial fullness and bloating, sensation of something sticking inside throat. There was no history of dysphasia, heartburn, gastrointestinal bleed, weight loss and no significant medical or surgical illness. Her physical and biochemical examination was normal. Upper gastrointestinal endoscopy showed normal esophagus with the presence of a single submucosal yellowish white elevated lesion, seen in the body of the stomach, measuring approximately 1 X 0.5 cm with normal overlying mucosa (Figure1A). This was followed by endoscopic ultrasonography (EUS) which revealed homogenous hypoechoic mass arising from submucosa of the stomach (Figure 1B). Biopsy was done and histology showed scanty tissue with normal stomach mucosa and submucosa showed a hyalinised area with presence of few scattered polygonal cells (Figure 2A). Hence an Immunohistochemistry was performed for S100 and CD68 which turned out to be strongly positive (Figure 4A and 4B). Thus, a diagnosis of GCT was made. Subsequently an endoscopic resection of the tumor was done. On gross, the resected nodule measured 1 X 0.5 X 0.5 cm and was yellowish white on cut surface. Histology showed normal gastric mucosa with underlying submucosa showing an unencapsulated mass composed of short fascicle polygonal to fusiform cells with abundant granular eosinophilic cytoplasm, positive on PAS and plump oval nuclei (Figure 3A and 3B). Mitotic figures and necrosis were not seen. This confirmed the diagnosis of GCT. On follow up patient got relieved of symptoms after resection and colonoscopy done later confirmed absence of GCT at any other site.
Discussion
GCT is an uncommon tumor, described for the first time by Abrikossoff in 1926. Though surface lesions (head, neck, trunk, extremities) are far more common than visceral (respiratory tract, biliary tract, genitourinary tract and gastrointestinal tract), any localisation is possible. In GIT stomach is rare site of involvement, most common being esophagus [1]. In 1995, Yasuda et al reported a case of gastric GCT with the mention of 24 cases previously reported in the literature [2]. From 1996 to 2006 Patti et al reviewed 6 more cases [1]. Since then, 11 more cases have been reported till 2014 [3]; but incidence of solitary gastric GCT is still very less and no exact literature available regarding the same. Gastric GCT shows synchronous and metachronous association. In at least 50% of cases gastric GCT is synchronously associated with esophageal GCT. While solitary gastric GCT is rare. Our case is of solitary GCT without any synchronous or metachronous association on follow up. Due to very few reported cases in literature, lesser data is available regarding exact age and sex of occurrence, it is stated that incidence is equal in both genders and can affect person of any age group. It is more common among black. Our patient was an adult female. Most of the gastric GCTs are benign, as in our case and are rarely malignant.
As gastric GCTs are usually asymptomatic, the majority of lesions are found incidentally during the investigation for other problems, just like our patient. Multifocal and large (i.e. > 1 cm) tumors may present with symptoms such as dysphasia and acid reflux, which were absent in our case.
Endoscopic ultrasonography has recently been used more frequently for determining the depth of tumor invasion in the GI wall, and may also be useful to evaluate GI tract submucosal tumors [4]. On EUS, GCTs usually arise in the lamina propria or deep mucosa layers of the GI tract, are usually less than 3 cm, hypoechoic, mildly inhomogeneous, and have smooth margins if benign [5]. Tada et al stressed that the treatment of choice for GCT should be determined by EUS findings [6]. When EUS shows that the tumor is localized in the submucosa and has not invaded the muscularis propria, endoscopy guided resection is treatment of choice as was done in our case which could avoid a major surgery.
Though EUS helps in narrowing the differential diagnosis of subepithelial lesions, the definitive diagnosis is always based on microscopic examination of the specimen. On Gross, GCTs is a nodular usually less than 2 cm in diameter grey to yellow white with cut surface being yellow [7]. On histology, it consists of large polygonal and fusiform cells disposed in compact nests which have abundant granular eosinophilic cytoplasm with central small round pyknotic nuclei [8]. Individualities of malignant GCTs are local recurrence, large size (>4 cm), rapid growth, invasion of adjacent organs and involvement of multiple layers in the gastrointestinal tract and are diagnosed with six histological criteria: necrosis, spindling, vesicular nuclei with large nucleoli, increased mitotic activity (>2 mitoses/10 high power fields), high nuclear to cytoplasmic ratio and nuclear pleomorphism. The various findings were similar in our case with no features of malignancy.
Immunohistochemical staining is positive for S-100, which supports Schwann cell origin of tumor and also for CD68, as in our case [9]. GCTs can also show immunoreactivity for vimentin, NSE and CD57, which were not done in our case due to nonavailability [10].
Recently, Parftt et al demonstrated expression of an intermediate filament protein called nestin (found normally in neuroectodermal stem cells and early skeletal muscle) in GCTs, some of which are located in the esophagus [11]. The expression of nestin in GCTs suggests that these tumors may arise from a common multipotential stem cell in the GI tract, which has the capability to differentiate along both interstitial cells of Cajal and peripheral nerve pathways. Nestin might be regarded as a useful marker for identifying GCTs [12].
There is controversy concerning the histogenesis of GCTs, thus several synonyms have been used to describe this entity. Myoblasts, Schwann cells, histiocytes, perineural fibroblasts and undifferentiated mesenchymal cells have been postulated as the origin of the tumor [13]. However, recent studies support a peripheral nerve-related cell of origin for the majority of these tumors based on the findings of cytoplasmic granules with numerous membrane-bound vacuoles containing myelin-like tubules and angulate bodies that show a close relation with pre-existent axons at the ultrastructural level, found between granular cells [13].
Conclusion
GCT of stomach is a rare tumor with its solitary occurrence being still rarer. Further, because of its deeper localisation, yielding scant tissue on biopsy, the diagnosis can be difficult can even be missed. Hence, a strong index of suspicion along with the immunohistochemistry is required for accurate diagnosis.
References
- Rosalia Patti et al. Granular cell tumor of stomach: A case report and review of literature. World J Gastroenterol 2006 June 7; 12(21): 3442-3445.
- Yasuda et al. Endoscopic removal of granular cell tumors. Gastrointest Endosc 1995; 41: 163-167.
- Dong Jun Kim et. al. A Case of Gastric Granular Cell Tumor: Review of Literature and Features of Endoscopic Ultrasonography The Korean Journal of Helicobacter and Upper Gastrointestinal Research, 2015; 15(1): 59-63.
- Nakachi A et al. Granular cell tumor of the rectum: a case report and review of the literature. J Gastroenterol. 2000; 35(8): 631-634.
- Palazzo L et al. Endosonographic features of esophageal granular cell tumors. Endoscopy. 1997; 29(9): 850-853.
- Tada S et al Polkowski M. Endoscopic ultrasound and endoscopic ultrasound-guided fine-needle biopsy for the diagnosis of malignant submucosal tumors. Endoscopy. 2005; 37(7): 635-645
- Eisen RN et al. Granular cell tumor of the biliary tree. A report of two cases and a review of the literature. Am J Surg Pathol. 1991; 15(5): 460-465.
- Weiss S et al. Enzinger and Weiss's Soft Tissue Tumors. 4th Edition. St Louis: CV Mosby; 2001.
- Wiech T et al. Histopathological classification of nonneoplastic and neoplastic gastrointestinal submucosal lesions. Endoscopy. 2005; 37(7): 630-634.
- Mazur MT et al. Granular cell tumor. Immunohistochemical analysis of 21 benign tumors and one malignant tumor. Arch Pathol Lab Med. 1990; 114(7): 692-696.
- Parfitt JR et al. Granular cell tumours of the gastrointestinal tract: expression of nestin and clinicopathological evaluation of 11 patients. Histopathology. 2006; 48(4): 424-430.
- Tada S et al. Granular cell tumor of the esophagus: endoscopic ultrasonographic demonstration and endoscopic removal. Am J Gastroenterol. 1990; 85(11): 1507-1511.
- Ordonez NG et al. Granular cell tumor: A review of the pathology and histogenesis. Ultrastruct Pathol. 1999; 23(4): 207-222.